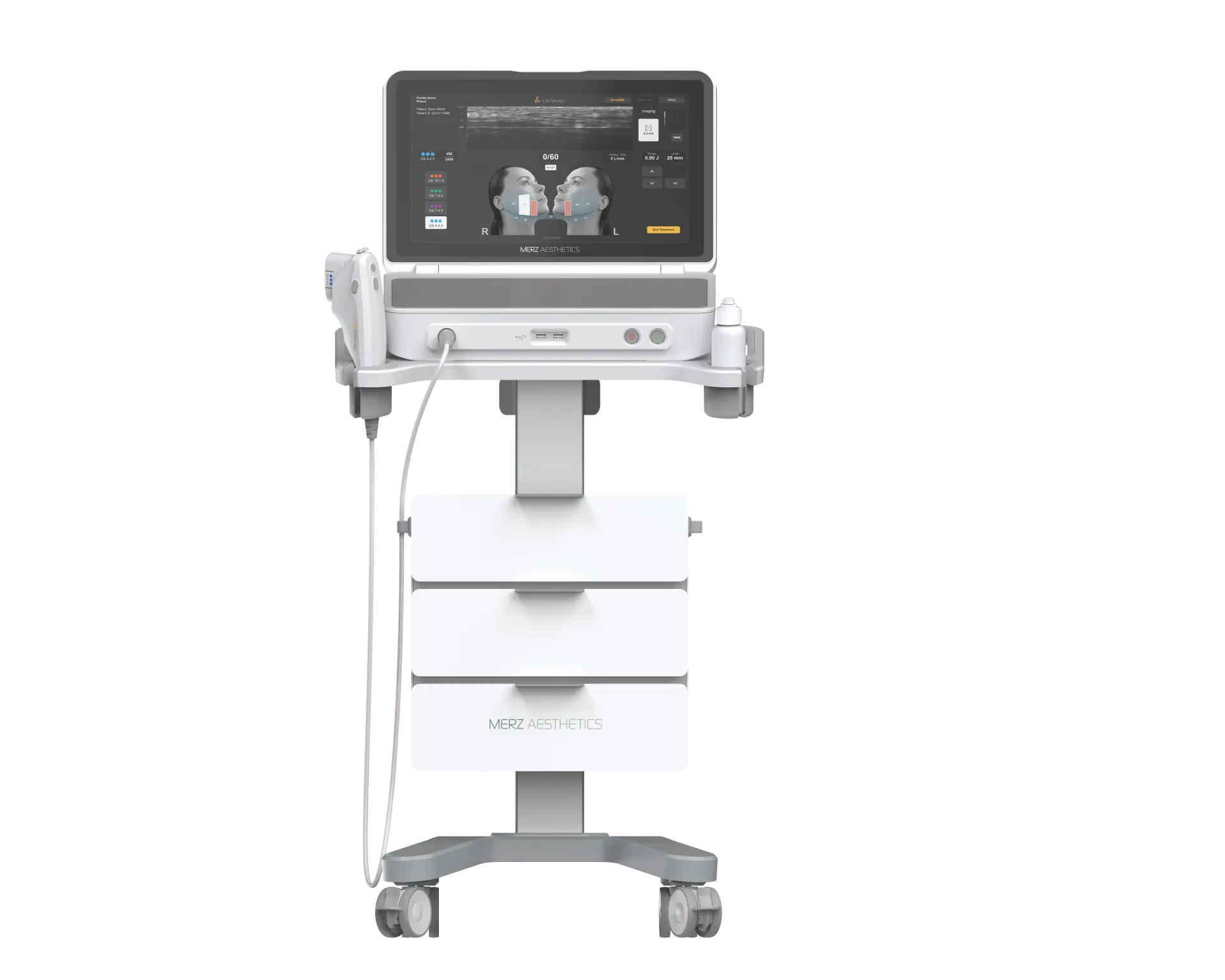
KFDA-Approved Devices Clinic Certified Medical Equipment in Gangnam Seoul
Every device at RE:BERRY Gangnam carries official KFDA (MFDS) approval and manufacturer certification, guaranteeing authentic equipment operated by licensed physicians. Our KFDA-approved devices clinic houses over 30 premium aesthetic platforms including Ultherapy Prime, Thermage FLX, Sofwave, Onda, Revinas, Soprano Titanium, and Potenza — each verified through official distributor channels with serial-number traceability. Treatments start from ₩19,000 at our 14,000 sq ft facility, 2 minutes from Gangnam Station Exit 10.
What Is a KFDA-Approved Devices Clinic?
A KFDA-approved devices clinic like RE:BERRY Gangnam Seoul operates 30+ medical aesthetic devices certified by Korea’s Ministry of Food and Drug Safety (MFDS/KFDA), ensuring authentic equipment with serial-number verification, manufacturer-trained physicians, and treatments from ₩19,000 — all within a 14,000 sq ft facility 2 minutes from Gangnam Station.
KFDA (Korea Food and Drug Administration, now officially MFDS — Ministry of Food and Drug Safety) approval is the Korean equivalent of US FDA clearance for medical devices. A KFDA-approved devices clinic exclusively uses equipment that has passed rigorous safety and efficacy testing by Korean regulatory authorities. This distinction matters because counterfeit and unregistered aesthetic devices are a documented problem in parts of Asia’s medical tourism market. At RE:BERRY Gangnam, Medical Director Dr. Yoon-Gon Ryu maintains direct relationships with official device distributors including Merz Aesthetics (Ultherapy), Solta Medical (Thermage FLX), Sofwave Medical, DEKA (Onda), Weyergans (Revinas), and Alma Lasers (Soprano Titanium). Every device in the clinic carries manufacturer certification displayed in the treatment room, and consumable tips/cartridges are verified authentic before each procedure. This KFDA-approved devices clinic model ensures patients receive the exact treatment parameters validated in clinical trials — not approximations from uncertified copies. Dr. Ryu and Dr. Yejin Oh are manufacturer-trained on each platform, holding individual operator certifications from device companies. RE:BERRY Gangnam’s 14,000 sq ft facility across B1-B2 of KI Tower dedicates separate treatment suites to lifting devices, skin rejuvenation platforms, and body contouring equipment. Our 8-language coordination team assists international patients who specifically seek verified KFDA-approved devices for their anti-aging treatments in Korea. The clinic operates 365 days a year from 10:00 AM to 8:00 PM, with same-day appointments available for patients who want to verify our certifications in person before committing to treatment.
Results from KFDA-Approved Devices at RE:BERRY Gangnam
Actual patient results using certified authentic devices at RE:BERRY Gangnam. Individual outcomes may vary.
How KFDA-Approved Devices Are Used at RE:BERRY Gangnam
A transparent, physician-performed protocol ensuring authentic device operation and optimal treatment outcomes.

Device Selection & Skin Analysis
Dr. Yoon-Gon Ryu begins every consultation with a comprehensive skin analysis using diagnostic imaging to assess your skin condition, laxity, fat distribution, and aging pattern. Based on this assessment, he selects the optimal KFDA-approved device or combination from RE:BERRY Gangnam’s portfolio of 30+ platforms. Each device addresses specific tissue depths and concerns: Ultherapy Prime targets the SMAS layer at 4.5mm, Thermage FLX delivers radiofrequency volumetrically through the dermis, and Sofwave concentrates SUPERB ultrasound at 1.5mm for mid-dermal collagen remodeling. This KFDA-approved devices selection process ensures the right technology for your specific goals.

Authenticity Verification
Before every treatment, our clinical team verifies the device’s authenticity and consumable status. For Thermage FLX, this means scanning the genuine Solta Medical tip serial number. For Ultherapy Prime, confirming the Merz-certified transducer count. For Sofwave, verifying the registered treatment cartridge. RE:BERRY Gangnam displays official KFDA-approved device certificates in each treatment room, and patients can request to see the device serial number and distributor invoice for any platform. This transparency protocol distinguishes a genuine KFDA-approved devices clinic from facilities using refurbished or counterfeit equipment. Our KFDA-approved devices are maintained on manufacturer-recommended service schedules with documented calibration records.
Physician-Operated Treatment
Every KFDA-approved device at RE:BERRY Gangnam is operated exclusively by Dr. Yoon-Gon Ryu or Dr. Yejin Oh — never delegated to nurses or technicians. This physician-operated model is critical because treatment outcomes depend heavily on operator expertise: energy settings, pass speed, treatment grid patterns, and real-time parameter adjustments during the session. Both physicians hold individual manufacturer certifications for each platform and have completed official training programs. Dr. Ryu’s KOL (Key Opinion Leader) status for Revinas and Juvelook reflects his recognized expertise with these KFDA-approved devices. Treatment duration varies from 15 minutes for targeted Botox to 90 minutes for full-face Ultherapy sessions.

Post-Treatment Documentation & Follow-Up
After every procedure, RE:BERRY Gangnam provides a detailed treatment record documenting the KFDA-approved device used, serial number, energy settings, shot count (for devices like Ultherapy and Revinas), and treatment area map. This documentation serves as both your medical record and verification that you received authentic treatment at certified parameters. Dr. Ryu schedules follow-up assessments at intervals appropriate to each device: 2 weeks for injectables, 4 weeks for energy-based devices, and 3 months for collagen-stimulating platforms. All documentation and aftercare instructions are provided in your preferred language through our 8-language team. Patients traveling from abroad receive digital copies of their KFDA-approved devices treatment records via secure messaging.
Why Choose a KFDA-Approved Devices Clinic in Gangnam
- Guaranteed Authenticity: Every device at RE:BERRY Gangnam’s KFDA-approved devices clinic carries serial-number traceability and manufacturer certification, eliminating the risk of counterfeit or refurbished equipment that plagues uncertified facilities.
- Clinical Trial Parameters: KFDA-approved devices deliver treatment at the exact energy levels and tissue depths validated in peer-reviewed clinical trials, ensuring you receive the outcomes supported by published evidence.
- Manufacturer-Trained Physicians: Dr. Yoon-Gon Ryu and Dr. Yejin Oh hold individual operator certifications from Merz, Solta, Sofwave, DEKA, Weyergans, and Alma Lasers — ensuring optimal device utilization.
- Comprehensive Device Portfolio: 30+ KFDA-approved platforms spanning lifting (Ultherapy, Thermage, Sofwave), skin rejuvenation (Potenza, LDM), body contouring (Onda, Berry Slim), and laser treatments — all under one roof.
- Safety Compliance: KFDA/MFDS certification means every device meets Korean medical safety standards for electromagnetic compatibility, electrical safety, and biocompatibility of patient-contact surfaces.
- Insurance and Warranty Coverage: Authentic KFDA-approved devices maintain full manufacturer warranty and are covered by RE:BERRY Gangnam’s medical liability insurance, providing financial protection for patients.
| Timeline | What to Expect |
|---|---|
| Consultation Day | Comprehensive skin analysis, device selection from 30+ KFDA-approved platforms, and treatment planning. Same-day treatment available for most procedures. |
| Treatment Day | Physician-operated treatment with verified authentic equipment. Duration ranges from 15 minutes (injectables) to 90 minutes (full-face lifting devices). |
| Week 1-2 | Initial results visible for injectable treatments and immediate-effect devices. Energy-based devices begin stimulating collagen production beneath the surface. |
| Month 1-2 | Progressive improvement as collagen remodeling from KFDA-approved lifting devices reaches peak biological response. Follow-up assessment with Dr. Ryu. |
| Month 3-6 | Full results achieved for collagen-stimulating devices (Thermage, Sofwave, Ultherapy). Maintenance treatment planning based on individual response. |
| Month 6-12 | Results maintained. Annual maintenance recommended for lifting devices. Skin booster and injectable treatments may be repeated at shorter intervals. |
KFDA-Approved Devices at RE:BERRY Gangnam: Key Platforms Compared
| Device | Ultherapy Prime | Thermage FLX | Sofwave | Onda | Revinas |
|---|---|---|---|---|---|
| Technology | Micro-focused ultrasound | Monopolar radiofrequency | SUPERB ultrasound | Coolwaves microwave | Pneumatic shock wave |
| Target Depth | 1.5-4.5mm (SMAS layer) | Full dermis volumetric | 1.5mm mid-dermis | Variable (fat + skin) | Connective tissue |
| KFDA Status | MFDS approved | MFDS approved | MFDS approved | MFDS approved | MFDS approved |
| Manufacturer Cert | Merz Aesthetics | Solta Medical | Sofwave Medical | DEKA | Weyergans |
| Price at RE:BERRY | ₩490,000/100 shots | ₩1,990,000/600 shots | ₩990,000/100 shots | ₩720,000 (40kJ+Titanium) | ₩1,790,000/6,000 shots |
| Treatment Time | 60-90 minutes | 45-60 minutes | 30-45 minutes | 30-40 minutes | 30-45 minutes |
| Results Duration | 12-18 months | 12-24 months | 6-12 months | 12-18 months | 6-12 months |
| Best For | Deep lifting, SMAS tightening | Skin tightening, collagen renewal | Fine lines, mild laxity | Body contouring + face lifting | Lymphatic drainage, tightening |
KFDA-Approved Device Treatment Pricing at RE:BERRY Gangnam
All prices include physician consultation. Non-covered medical service. Prices valid April-June 2026.
Lifting Device Essentials
- Inmode FX 10 minutes (₩190,000)
- Ultherapy Prime 100 shots (₩490,000)
- Sofwave 100 shots (₩990,000)
- Physician-operated with certified device
- Authenticity verification before treatment
Signature Lifting Package
- Ultherapy 530 shots
- Sofwave 200 shots
- Onda lifting 100kJ
- Revinas 12,000 shots
- LDM aftercare session
- All KFDA-approved devices, physician-operated
Thermage FLX 600 Shots
- 600 shots full-face treatment
- Genuine Solta Medical certified tip
- Serial number verification included
- Physician-operated by Dr. Ryu or Dr. Oh
- 3-month follow-up assessment
Meet Your Doctor

Dr. Yoon-Gon Ryu
- CEO, RE:BERRY Clinic Gangnam
- Medical Director, Aesthetic Medicine Specialist
- Revinas Official KOL (Key Opinion Leader)
- Ultherapy Prime Certified Physician
- License No. 131644
Medically reviewed by Dr. Yoon-Gon Ryu, CEO / Medical Director, RE:BERRY
The Science Behind KFDA-Approved Aesthetic Devices
Peer-reviewed evidence supporting the efficacy of MFDS-certified aesthetic platforms used at RE:BERRY Gangnam.
Micro-Focused Ultrasound (Ultherapy) Clinical Evidence
Micro-focused ultrasound with visualization (MFU-V), marketed as Ultherapy, received KFDA/MFDS approval based on extensive clinical evidence demonstrating its ability to target the SMAS (superficial musculoaponeurotic system) layer non-invasively. The device creates precise thermal coagulation points at depths of 1.5mm, 3.0mm, and 4.5mm, triggering neocollagenesis and tissue contraction. The KFDA-approved Ultherapy Prime at RE:BERRY Gangnam delivers calibrated ultrasound energy at standardized focal depths, a parameter that cannot be replicated by counterfeit devices. Ultherapy Prime treatment at RE:BERRY Gangnam starts from ₩490,000 per 100 shots, with physician-operated precision ensuring optimal energy delivery to the target tissue plane.
Monopolar Radiofrequency (Thermage FLX) Mechanism
Thermage FLX uses monopolar radiofrequency energy to deliver volumetric heating throughout the full thickness of the dermis. The KFDA-approved device’s AccuREP technology measures tissue impedance 100 times per second, automatically adjusting energy delivery for uniform heating. This real-time feedback system is embedded in the genuine Solta Medical platform and cannot be replicated by aftermarket devices. Clinical studies confirm that authentic Thermage FLX treatment produces measurable increases in dermal collagen density at 6-month follow-up, with tissue tightening effects lasting 12-24 months in properly treated patients.
SUPERB Ultrasound Technology (Sofwave)
Sofwave’s Synchronous Ultrasound Parallel Beam Technology (SUPERB) concentrates ultrasound energy at a fixed depth of 1.5mm in the mid-dermis, the zone of maximum collagen density. The KFDA-approved platform includes Sofcool integrated cooling that protects the epidermis while delivering therapeutic temperatures of 60-70 degrees Celsius to the target zone. This precise depth targeting distinguishes authentic Sofwave from generic ultrasound devices and explains why MFDS certification specifically validates the SUPERB delivery mechanism. At RE:BERRY Gangnam, Sofwave treatment is physician-operated using verified cartridges from Sofwave Medical’s official Korean distributor.

Regulatory Framework: KFDA/MFDS Device Approval Process
Korea’s MFDS (formerly KFDA) classifies medical aesthetic devices into risk categories (Class I-IV), with higher-energy platforms like Ultherapy and Thermage requiring Class III or IV approval. The approval process mandates submission of manufacturing quality system documentation (ISO 13485), pre-clinical safety data (biocompatibility, electromagnetic compatibility, electrical safety testing), and clinical evidence demonstrating efficacy for the claimed indication. Post-market surveillance requirements ensure ongoing safety monitoring. This regulatory framework means KFDA-approved devices at certified clinics like RE:BERRY Gangnam meet internationally recognized safety standards comparable to FDA 510(k) clearance in the United States and CE marking in Europe.
Fabi SG et al. Evaluation of microfocused ultrasound with visualization for lifting, tightening, and wrinkle reduction of the decolletage. J Am Acad Dermatol. 2013;69(6):965-971. doi:10.1016/j.jaad.2013.06.045
Suh DH et al. Monopolar radiofrequency treatment for facial laxity: a systematic review and meta-analysis. Dermatol Ther. 2020;33(6):e14270. doi:10.1111/dth.14270
Halachmi S et al. Treatment of wrinkles and skin tightening using Sofwave’s novel SUPERB technology. J Drugs Dermatol. 2020;19(11):1116-1120. doi:10.36849/JDD.2020.5512
What Our Patients Say About KFDA-Approved Devices at RE:BERRY Gangnam
“I specifically chose RE:BERRY Gangnam because they could show me the Thermage FLX certification and genuine Solta tip serial number before my treatment. After hearing horror stories about counterfeit tips at cheaper clinics, the transparency was worth every won. My results at 3 months were exactly what the clinical studies promised.”
“Dr. Ryu walked me through every KFDA-approved device he recommended for my combination treatment. He showed me the Ultherapy certification on the wall and the Revinas KOL plaque. I received 200 shots of Ultherapy and 6,000 Revinas in one session. The lifting results at 6 weeks were remarkable.”
“As a repeat visitor to Korean clinics, I have seen facilities that cut corners on equipment authenticity. RE:BERRY Gangnam is the real deal — 30+ genuine devices, all with visible certifications, operated by the doctors themselves. The Sofwave treatment I received for my jawline gave visible tightening within 4 weeks.”
Frequently Asked Questions About KFDA-Approved Devices Clinic
Expert answers from RE:BERRY Gangnam’s device specialists.
30+ KFDA-approved devices across lifting, skin rejuvenation, body contouring, and laser categories. Key platforms include Ultherapy Prime (Merz), Thermage FLX (Solta), Sofwave, Onda (DEKA), Revinas (Weyergans), Soprano Titanium (Alma), Potenza, and LDM.
KFDA and MFDS refer to the same regulatory body. Korea’s Food and Drug Administration was officially renamed to the Ministry of Food and Drug Safety (MFDS) in 2013. Both terms indicate identical regulatory approval. RE:BERRY Gangnam’s devices carry current MFDS certification.
3 verification methods: (1) Ask to see the manufacturer certification displayed in the treatment room, (2) Request the device serial number and cross-check with the manufacturer’s Korean distributor, and (3) For consumables like Thermage tips, check the holographic seal and serial number. RE:BERRY Gangnam provides all 3 verifications on request.
₩1,990,000 for authentic Thermage FLX 600 shots at RE:BERRY Gangnam versus ₩500,000-800,000 at uncertified facilities reflects genuine consumable costs. Each Thermage tip costs the clinic approximately ₩600,000 wholesale. Clinics offering dramatically lower prices may use counterfeit tips that deliver inconsistent energy.
Ultherapy Prime is the most effective single device for severe laxity, targeting the SMAS layer at 4.5mm depth. For maximum results, RE:BERRY Gangnam’s Signature Lifting Package (₩5,990,000) combines Ultherapy 530 shots + Sofwave 200 shots + Onda 100kJ + Revinas 12,000 shots across multiple tissue depths.
Yes, RE:BERRY Gangnam maintains current-generation platforms: Thermage FLX (4th gen, not older CPT), Ultherapy Prime (updated imaging), Sofwave with Sofcool, and Onda with latest Coolwaves handpieces. Devices are upgraded when manufacturers release new KFDA-approved generations.
Yes, 65% of lifting patients at RE:BERRY Gangnam receive multi-device combination treatments. Common combinations include Thermage FLX + Ultherapy for different tissue depths, Sofwave + Onda for face and body, and Potenza + skin boosters for texture improvement.
₩19,000 for wrinkle Botox (entry-level), ₩190,000 for Inmode FX, ₩490,000 for Ultherapy 100 shots, ₩990,000 for Sofwave 100 shots, ₩1,990,000 for Thermage 600 shots. Signature Lifting Package combining 4 devices is ₩5,990,000.
Yes, Korean media investigations have documented cases of counterfeit Thermage tips and unauthorized device copies in budget clinics. The MFDS reports approximately 3-5% of inspected facilities have compliance issues. Choosing a verified KFDA-approved devices clinic like RE:BERRY Gangnam eliminates this risk.
7 official manufacturer certifications: Ultherapy Prime (Merz Aesthetics), Sofwave (Sofwave Medical), Revinas KOL (Weyergans), Onda Premium (DEKA), Thermage FLX (Solta Medical), Soprano Titanium (Alma Lasers), and Volnewmer (Jeisys Medical). All displayed in the clinic.
100% authentic Solta Medical Thermage FLX with verifiable serial number. RE:BERRY Gangnam is an official certified clinic for Thermage FLX. Every treatment uses genuine Solta tips with holographic authentication seals, verified before the patient’s treatment begins.
KFDA/MFDS approval is equivalent to US FDA 510(k) clearance in rigor. Both require clinical evidence, manufacturing quality documentation (ISO 13485), and post-market surveillance. Many KFDA-approved devices at RE:BERRY Gangnam also hold US FDA and European CE certifications.
Yes. RE:BERRY Gangnam displays all manufacturer certifications in treatment rooms. You can request to see device serial numbers, distributor invoices, and consumable authentication seals before any procedure. Same-day consultation visits for certification verification are available 365 days.
Onda Coolwaves (₩1,090,000 for 100kJ body) is the primary KFDA-approved body contouring device at RE:BERRY Gangnam. It targets adipose tissue with microwave energy at a specific frequency that selectively heats fat cells while protecting surrounding tissue. Body Thermage is also available for skin tightening.
Yes, the KFDA-approved Revinas at RE:BERRY Gangnam delivers precisely measured pneumatic shock wave pulses. The Signature Lifting Package includes 12,000 shots. Dr. Yoon-Gon Ryu holds Revinas KOL (Key Opinion Leader) certification from Weyergans, reflecting his expertise with this KFDA-approved device.
12-18 months for Ultherapy and Thermage maintenance, 6-12 months for Sofwave and Revinas. The exact interval depends on your individual collagen response and aging pattern. Dr. Ryu creates a personalized maintenance schedule during your 3-month follow-up assessment.
Yes, you can fly the same day after most energy-based device treatments. There are no altitude restrictions for ultrasound, radiofrequency, or microwave-based devices. Minor redness or warmth typically resolves within 2-6 hours.
Yes, RE:BERRY Gangnam provides device consultation and aftercare in 8 languages: English, Japanese, Chinese (Simplified and Traditional), Thai, Vietnamese, Mongolian, and Farsi. Our multilingual coordinators explain each KFDA-approved device and its certification in your language.
1-2 days for most patients. Single-device treatments require 1 day with same-day availability. Multi-device combination packages like the Signature Lifting Package (₩5,990,000) can be completed in a single 2-3 hour session. A follow-up visit at 2 weeks is optional if you are still in Seoul.
Sofwave with Sofcool integrated cooling and the latest Revinas handpiece iteration are among the most recently certified additions to RE:BERRY Gangnam’s KFDA-approved devices portfolio. The clinic updates equipment when manufacturers release new MFDS-approved generations.
No medical treatment guarantees specific results. However, KFDA-approved devices deliver validated energy parameters that produce outcomes consistent with published clinical trial data. RE:BERRY Gangnam’s physician-operated model and authentic equipment maximize the probability of achieving study-demonstrated results.
Yes, the KFDA-approved Soprano Titanium (Alma Lasers) at RE:BERRY Gangnam is certified for Fitzpatrick skin types I-VI. Its in-motion SHR technology with simultaneous contact cooling makes it safe for darker skin types that are contraindicated for older laser hair removal platforms.
3 key reasons: (1) 30+ KFDA-approved devices with manufacturer certifications displayed and serial numbers verifiable; (2) All treatments physician-operated by Dr. Yoon-Gon Ryu (Revinas/Juvelook KOL) or Dr. Yejin Oh; (3) Located 2 minutes from Gangnam Station with 8-language support for international patients.
RE:BERRY Gangnam’s KFDA-approved approach is distinguished by complete transparency: manufacturer certifications displayed, serial numbers available on request, genuine consumables verified before each treatment, and physician-only operation with no delegation to nurses or technicians.
Yes, the KFDA-approved Potenza RF microneedling device at RE:BERRY Gangnam effectively treats acne scars by delivering radiofrequency energy through insulated or non-insulated microneedles. Pricing starts at ₩490,000 combined with Juvelook skin booster. Multiple tip configurations allow treatment customization for different scar types.
Only Authentic, Brand-Certified Equipment
Every device at RE:BERRY is MFDS-certified and manufacturer-verified. We never use counterfeit or grey-market equipment.
Experience Genuine KFDA-Approved Devices at RE:BERRY Gangnam
Book your consultation with Dr. Yoon-Gon Ryu or Dr. Yejin Oh to discuss treatment with certified, authentic KFDA-approved devices. 30+ platforms, physician-operated, 2 minutes from Gangnam Station Exit 10.
Book Your Device Consultation
Fill out the form and our multilingual team will confirm within 2 hours via your preferred contact method.
- ✓ Free consultation with Dr. Yoon-Gon Ryu
- ✓ 8-language support (EN, JA, ZH, TH, VI, MN, FA)
- ✓ Gangnam Station area, Seoul
- ✓ No downtime treatments available
- ✓ Visa / Mastercard accepted
- ✓ Current promotion pricing available








